Expo
view channel
view channel
view channel
view channel
view channel
view channel
view channel
view channel
view channel
Clinical Chem.Molecular DiagnosticsHematology
MicrobiologyPathologyTechnologyIndustry
Events
Webinars

- Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
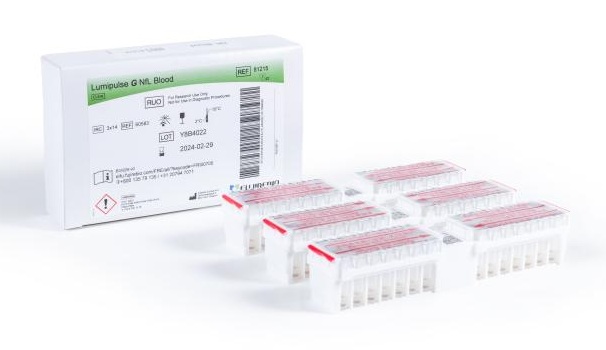
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- CSF Biomarker Improves Diagnosis of Parkinson’s Disease and Lewy Body Dementia
- Simple Urine Home Test Kit Could Detect Early-Stage Breast Cancer
- Plasma ctDNA Testing Predicts Breast Cancer Recurrence After Neoadjuvant Therapy
- New Respiratory Panel Expands Pathogen Detection to 25 Targets
- Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
- Blood Biomarker Predicts Cognitive Outcomes After Cardiac Arrest
- Liquid Biopsy Enables Faster Diagnosis of Childhood Cancer in Africa
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Fast and Easy Test Could Revolutionize Blood Transfusions
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
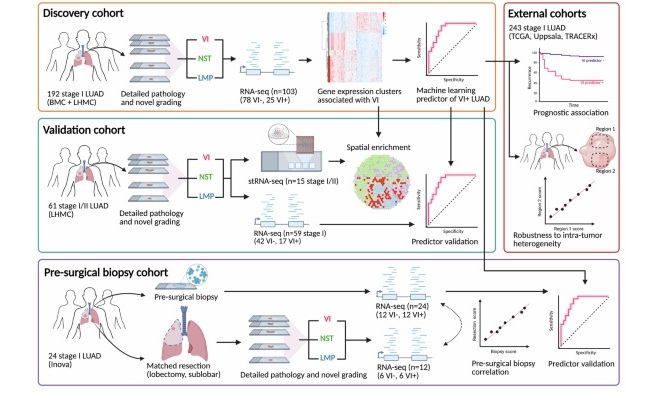
- Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
- Cancer Mutation ‘Fingerprints’ to Improve Prediction of Immunotherapy Response
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
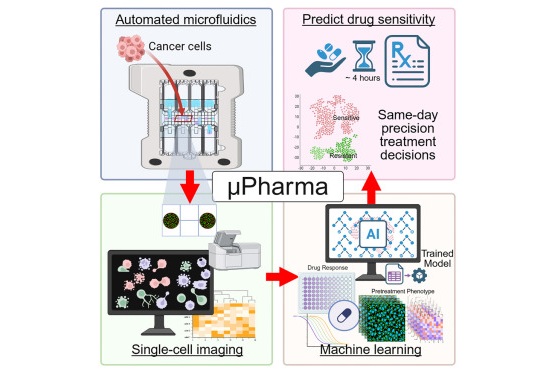
- Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
- Online Tool Supports Family Screening for Inherited Cancer Risk
- Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
- New Electronic Pipette Enhances Workflows with Touchscreen Control
- Co-Diagnostics Agreement Expands Commercial and Distribution Reach in South Asia
- Automated MSI Test Gains IVDR Certification to Guide CRC Therapy
- New Partnership Brings Alzheimer’s Blood Biomarker Test to Community Screening Network
- MGI Tech Strengthens Sequencing Portfolio with Dual Acquisition
- Agilent Technologies Acquires Pathology Diagnostics Company Biocare Medical
- Study Reveals Diagnostic and Therapeutic Target in Rare Pancreatic Tumors
- Large-Scale Study Maps DNA Damage Signatures Across Multiple Cancers
- Study Identifies Distinct Immune Signatures to Early Depression and Psychosis
- Genetic Mutation Behind Aggressive Adult Leukemia Offers Treatment Clues
- Disease Gene Discovery Advances Diagnosis of Rare Movement Disorders
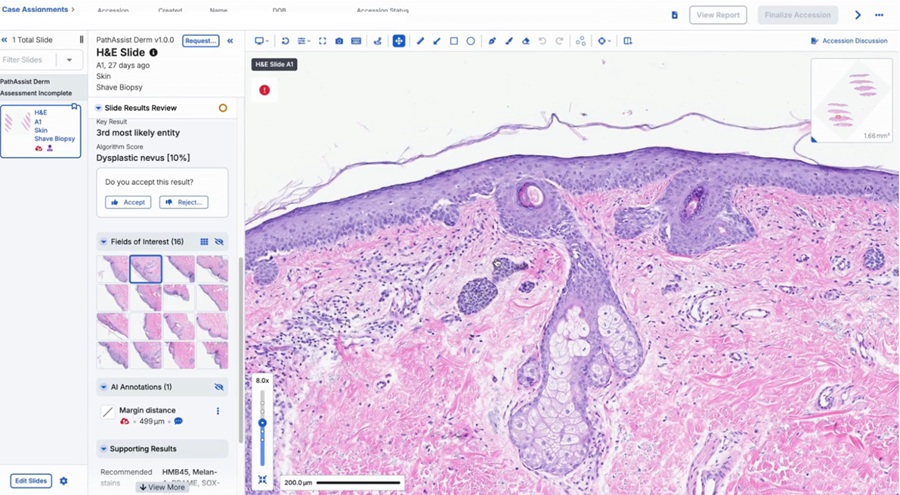
- AI-Powered Tool to Transform Dermatopathology Workflow
- New Chromogenic Culture Media Enable Rapid Detection of Candida Infections
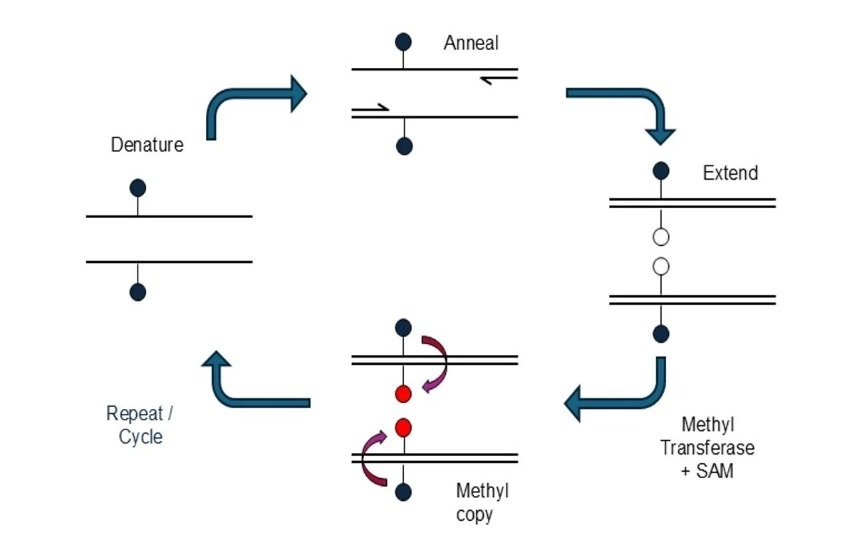
- Novel mcPCR Technology to Transform Testing of Clinical Samples
- Sex Differences in Alzheimer’s Biomarkers Linked to Faster Cognitive Decline
- AI Tool Predicts Chemotherapy Response from Biopsy Slides

 Expo
Expo
- Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- CSF Biomarker Improves Diagnosis of Parkinson’s Disease and Lewy Body Dementia
- Simple Urine Home Test Kit Could Detect Early-Stage Breast Cancer
- Plasma ctDNA Testing Predicts Breast Cancer Recurrence After Neoadjuvant Therapy
- New Respiratory Panel Expands Pathogen Detection to 25 Targets
- Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
- Blood Biomarker Predicts Cognitive Outcomes After Cardiac Arrest
- Liquid Biopsy Enables Faster Diagnosis of Childhood Cancer in Africa
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Fast and Easy Test Could Revolutionize Blood Transfusions
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
- Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
- Cancer Mutation ‘Fingerprints’ to Improve Prediction of Immunotherapy Response
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
- Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
- Online Tool Supports Family Screening for Inherited Cancer Risk
- Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
- New Electronic Pipette Enhances Workflows with Touchscreen Control
- Co-Diagnostics Agreement Expands Commercial and Distribution Reach in South Asia
- Automated MSI Test Gains IVDR Certification to Guide CRC Therapy
- New Partnership Brings Alzheimer’s Blood Biomarker Test to Community Screening Network
- MGI Tech Strengthens Sequencing Portfolio with Dual Acquisition
- Agilent Technologies Acquires Pathology Diagnostics Company Biocare Medical
- Study Reveals Diagnostic and Therapeutic Target in Rare Pancreatic Tumors
- Large-Scale Study Maps DNA Damage Signatures Across Multiple Cancers
- Study Identifies Distinct Immune Signatures to Early Depression and Psychosis
- Genetic Mutation Behind Aggressive Adult Leukemia Offers Treatment Clues
- Disease Gene Discovery Advances Diagnosis of Rare Movement Disorders
- AI-Powered Tool to Transform Dermatopathology Workflow
- New Chromogenic Culture Media Enable Rapid Detection of Candida Infections
- Novel mcPCR Technology to Transform Testing of Clinical Samples
- Sex Differences in Alzheimer’s Biomarkers Linked to Faster Cognitive Decline
- AI Tool Predicts Chemotherapy Response from Biopsy Slides