Expo
view channel
view channel
view channel
view channel
view channel
view channel
view channel
view channel
view channel
Clinical Chem.Molecular DiagnosticsHematologyImmunology
PathologyTechnologyIndustry
Events
Webinars

- Routine Blood Markers Predict Heart Failure Risk in Prediabetes
- First IVD Immunoassay to Detect Alzheimer’s Risk Gene Variant Receives CE Mark
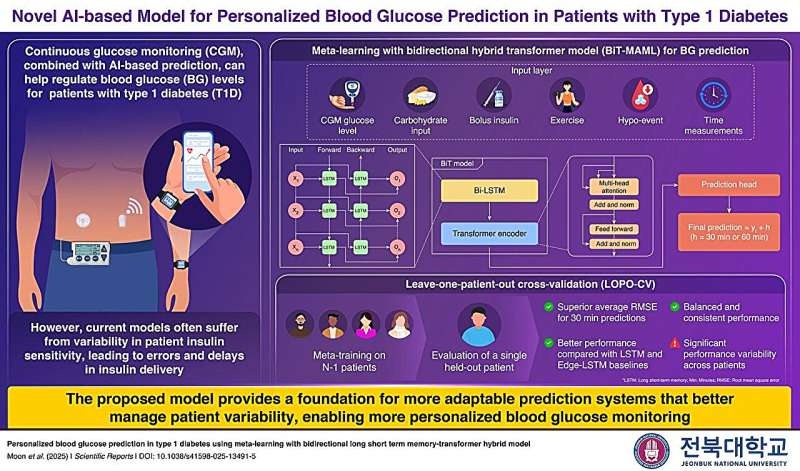
- AI Model Enables Personalized Glucose Predictions for Type 1 Diabetes
- AI-Powered Blood Test Distinguishes Deadly Cardiac Events
- Blood Test Tracks Transplant Health Using Donor DNA
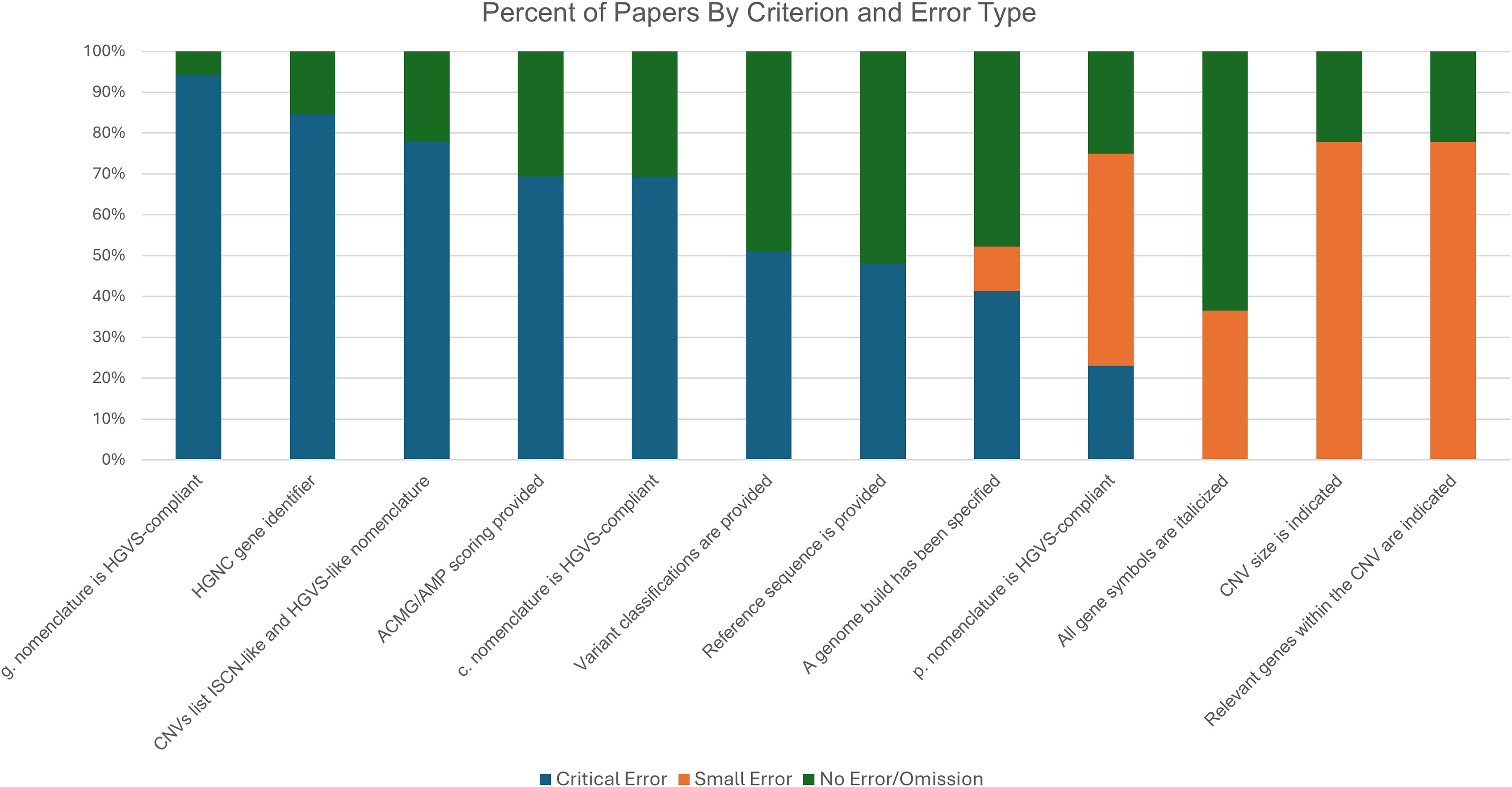
- Study Reveals Widespread Errors in Gene Variant Naming
- New Biomarkers Indicate Higher Liver Cancer Risk in Chronic Hepatitis B Patients
- Rapid Host-Response Test Distinguishes Bacterial and Viral Infections in Minutes
- Precision Analyzer Reveals ‘Chameleon Proteins’ Causing Intractable Diseases
- Alzheimer's Blood Marker Could Improve Detection of Heart and Kidney Diseases
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Fast and Easy Test Could Revolutionize Blood Transfusions
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
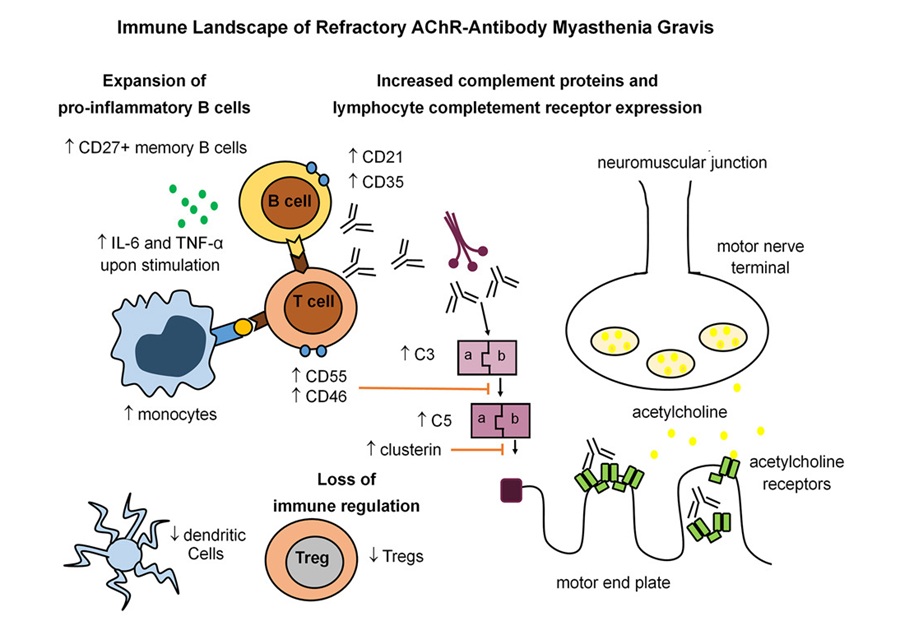
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Blood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
- Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
- Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Blood-Based Viral Signature Identified in Crohn’s Disease
- Hidden Gut Viruses Linked to Colorectal Cancer Risk
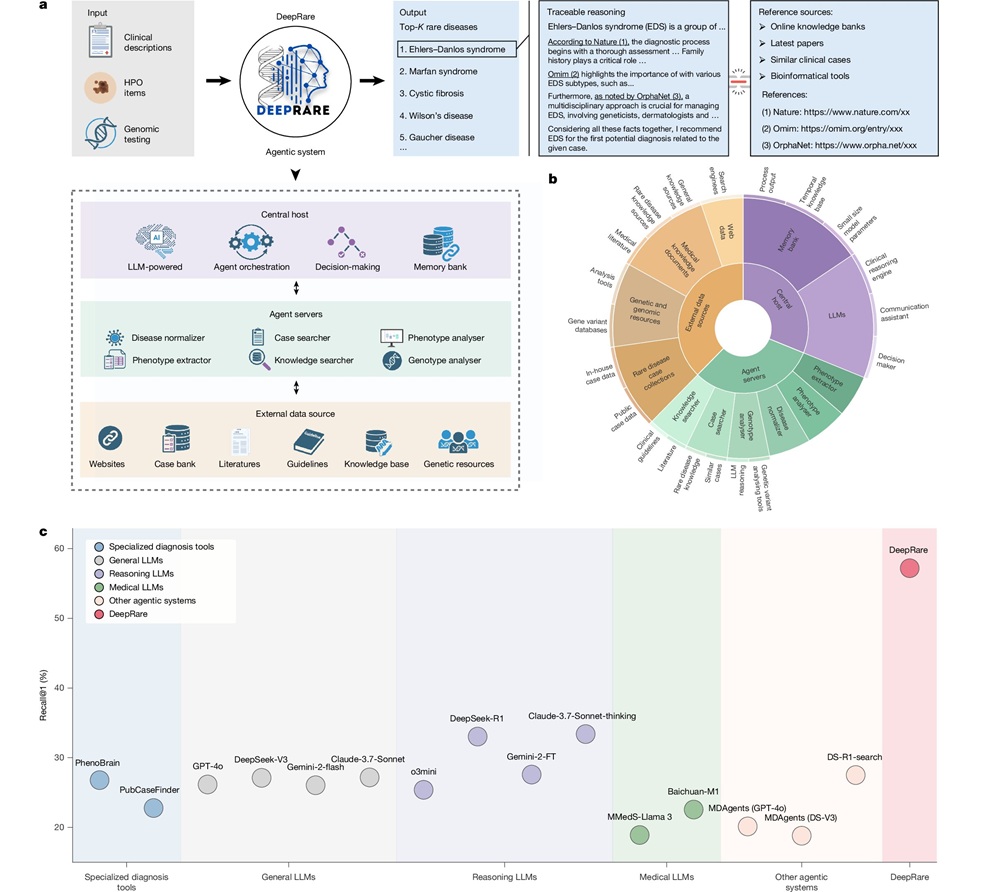
- AI Model Outperforms Clinicians in Rare Disease Detection
- AI-Driven Diagnostic Demonstrates High Accuracy in Detecting Periprosthetic Joint Infection
- Blood Test “Clocks” Predict Start of Alzheimer’s Symptoms
- AI-Powered Biomarker Predicts Liver Cancer Risk
- Robotic Technology Unveiled for Automated Diagnostic Blood Draws
- New Partnership Brings Alzheimer’s Blood Biomarker Test to Community Screening Network
- MGI Tech Strengthens Sequencing Portfolio with Dual Acquisition
- Agilent Technologies Acquires Pathology Diagnostics Company Biocare Medical
- Cepheid Joins CDC Initiative to Strengthen U.S. Pandemic Testing Preparednesss
- QuidelOrtho Collaborates with Lifotronic to Expand Global Immunoassay Portfolio
- Genetic Discovery Could Improve Diagnosis of Drug-Resistant Epilepsy
- Genetic Discovery May Improve Diagnosis of Rare Dementia Subtype
- Mass Spectrometry Technique Detects Protein and Sugar Changes in Neurodegeneration
- Barcoded DNA Sheds Light on Hidden Complexities in Breast Cancer Detection
- CRISPR-Based Platform Pinpoints Drivers of Acute Myeloid Leukemia in Patient Cells
- New Chromogenic Culture Media Enable Rapid Detection of Candida Infections
- Novel mcPCR Technology to Transform Testing of Clinical Samples
- Sex Differences in Alzheimer’s Biomarkers Linked to Faster Cognitive Decline
- AI Tool Predicts Chemotherapy Response from Biopsy Slides
- World’s First Optical Microneedle Device to Enable Blood-Sampling-Free Clinical Testing

 Expo
Expo
- Routine Blood Markers Predict Heart Failure Risk in Prediabetes
- First IVD Immunoassay to Detect Alzheimer’s Risk Gene Variant Receives CE Mark
- AI Model Enables Personalized Glucose Predictions for Type 1 Diabetes
- AI-Powered Blood Test Distinguishes Deadly Cardiac Events
- Blood Test Tracks Transplant Health Using Donor DNA
- Study Reveals Widespread Errors in Gene Variant Naming
- New Biomarkers Indicate Higher Liver Cancer Risk in Chronic Hepatitis B Patients
- Rapid Host-Response Test Distinguishes Bacterial and Viral Infections in Minutes
- Precision Analyzer Reveals ‘Chameleon Proteins’ Causing Intractable Diseases
- Alzheimer's Blood Marker Could Improve Detection of Heart and Kidney Diseases
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Fast and Easy Test Could Revolutionize Blood Transfusions
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Blood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
- Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
- Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Blood-Based Viral Signature Identified in Crohn’s Disease
- Hidden Gut Viruses Linked to Colorectal Cancer Risk
- AI Model Outperforms Clinicians in Rare Disease Detection
- AI-Driven Diagnostic Demonstrates High Accuracy in Detecting Periprosthetic Joint Infection
- Blood Test “Clocks” Predict Start of Alzheimer’s Symptoms
- AI-Powered Biomarker Predicts Liver Cancer Risk
- Robotic Technology Unveiled for Automated Diagnostic Blood Draws
- New Partnership Brings Alzheimer’s Blood Biomarker Test to Community Screening Network
- MGI Tech Strengthens Sequencing Portfolio with Dual Acquisition
- Agilent Technologies Acquires Pathology Diagnostics Company Biocare Medical
- Cepheid Joins CDC Initiative to Strengthen U.S. Pandemic Testing Preparednesss
- QuidelOrtho Collaborates with Lifotronic to Expand Global Immunoassay Portfolio
- Genetic Discovery Could Improve Diagnosis of Drug-Resistant Epilepsy
- Genetic Discovery May Improve Diagnosis of Rare Dementia Subtype
- Mass Spectrometry Technique Detects Protein and Sugar Changes in Neurodegeneration
- Barcoded DNA Sheds Light on Hidden Complexities in Breast Cancer Detection
- CRISPR-Based Platform Pinpoints Drivers of Acute Myeloid Leukemia in Patient Cells
- New Chromogenic Culture Media Enable Rapid Detection of Candida Infections
- Novel mcPCR Technology to Transform Testing of Clinical Samples
- Sex Differences in Alzheimer’s Biomarkers Linked to Faster Cognitive Decline
- AI Tool Predicts Chemotherapy Response from Biopsy Slides
- World’s First Optical Microneedle Device to Enable Blood-Sampling-Free Clinical Testing